The result was an inverse energy distribution, which is characteristic of negative temperatures. They made the atoms attract and adjusted the lasers to change the atoms’ energy levels, making the majority of them high-energy, and so flipping the valley into an energy hill. Schneider’s team then turned this positive temperature system negative by doing two things. The atoms were also made to repel each other to ensure they remained fixed in place. 
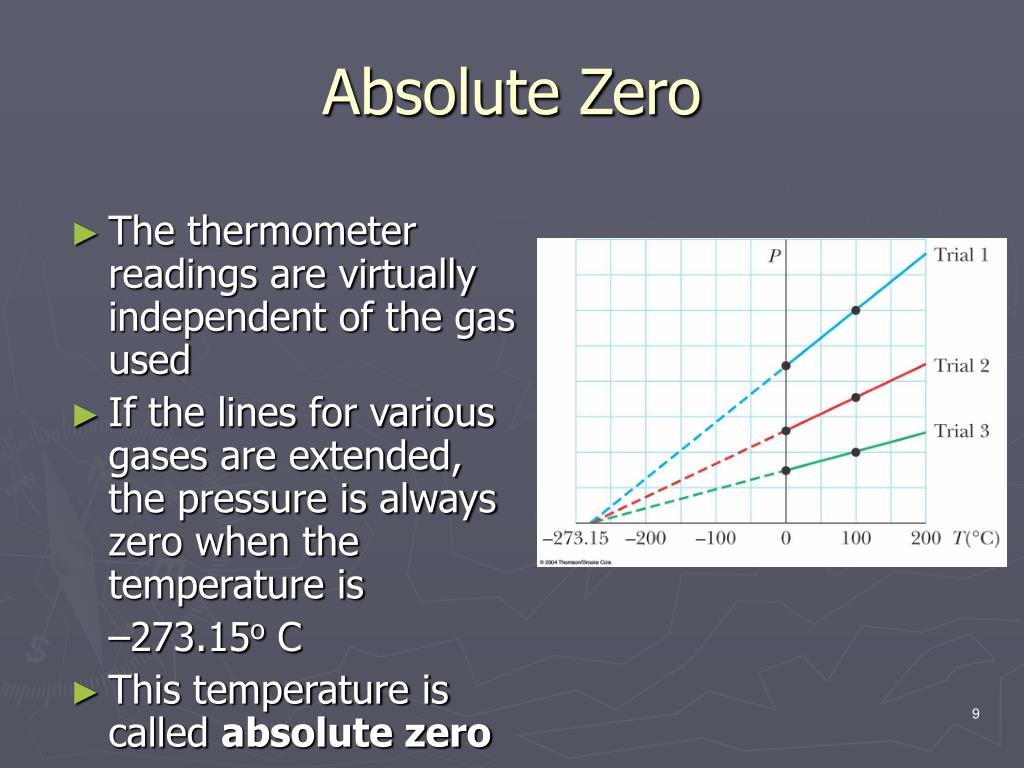
They then used lasers to place the atoms along the curve of an energy valley with the majority of the atoms in lower energy states. To enter the negative realm, Schneider and his colleagues began by cooling atoms to a fraction above absolute zero and placing them in a vacuum. “The temperature scale as we know it starts at zero and goes up to infinity, but it doesn’t stop there,” says Ulrich Schneider of the Ludwig Maximilian University of Munich in Germany. The resulting thermometer is mind-bending with a scale that starts at zero, ramps up to plus infinity, then jumps to minus infinity before increasing through the negative numbers until it reaches negative absolute zero, which corresponds to all particles sitting at the top of the energy hill. The end of this negative scale is reached when all particles are at the top of the energy hill. Because this breaks the energy-entropy correlation, it marks the start of the negative temperature scale, where the distribution of energies is reversed – instead of most particles having a low energy and a few having a high, most have a high energy and just a few have a low energy. In principle, though, it should be possible to keep heating the particles up, while driving their entropy down. As a result, this point represents the end of the positive temperature scale. Increase the energy any further and you’d start to lower the entropy again, because the particles wouldn’t be evenly spread. This would be an equal number of particles at every point on the landscape. 
Spread along the side of the hill, now the particles have different energies, so entropy is higher.Īccording to temperature’s entropic definition, the highest positive temperature possible corresponds to the most disordered state of the system. At absolute zero, particles are motionless and all have no energy so are all at the bottom of the valley, giving a minimum entropy.Īs the gas heats up, the average energy of the particles increases, with some gaining lots of extra energy but most just a small amount. Picture a hill next to a valley (see image) with the height of the landscape corresponding to the energy of a particle – and the chance of finding a particle at a certain height representing entropy. Energy landscapeĪbove absolute zero, adding more energy corresponds to an increase in entropy. Temperature also depends on the way in which the particle energies are distributed within the gas, which determines their entropy, or disorder. That does not tell the whole story, however.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
