|
When an electron is added to such a small atom, increased electron–electron repulsions tend to destabilize the anion. In contrast to the chemistry of the second-period elements, the chemistry of the third-period elements is more representative of the chemistry of the respective group.ĭue to their small radii, second-period elements have electron affinities that are less negative than would be predicted from general periodic trends. The anomalous chemistry of second-period elements results from three important characteristics: small radii, energetically unavailable d orbitals, and a tendency to form pi (π) bonds with other atoms. Consequently, the elements of the third period (n = 3: Na, Mg, Al, Si, P, S, and Cl) are generally more representative of the group to which they belong. The chemistry of the second-period element of each group (n = 2: Li, Be, B, C, N, O, and F) differs in many important respects from that of the heavier members, or congeners, of the group. Unique Chemistry of the Lightest Elements Thus in the third row of the periodic table, the largest increase in ionization energy corresponds to removing the fourth electron from (Al), the fifth electron from Si, and so forththat is, removing an electron from an ion that has the valence electron configuration of the preceding noble gas. 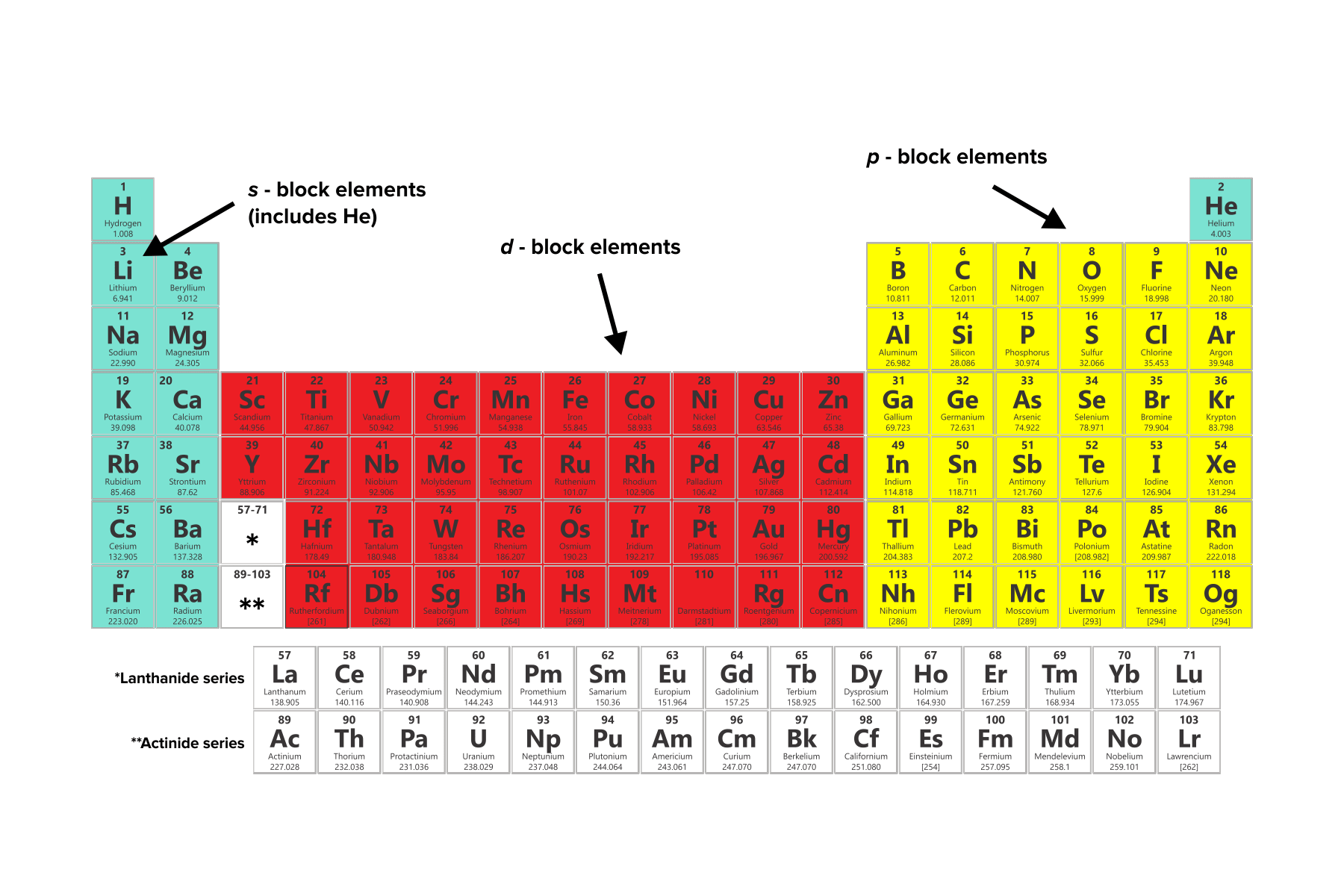
The semimetals lie along the diagonal line separating the metals from the nonmetals and exhibit intermediate properties. Consequently, the elements in the upper right of the periodic table are the smallest and most electronegative the elements in the bottom left are the largest and least electronegative. In contrast, atomic size decreases from left to right and from bottom to top. Ionization energies, the magnitude of electron affinities, and electronegativities generally increase from left to right and from bottom to top. The table shows the generic valence electron. 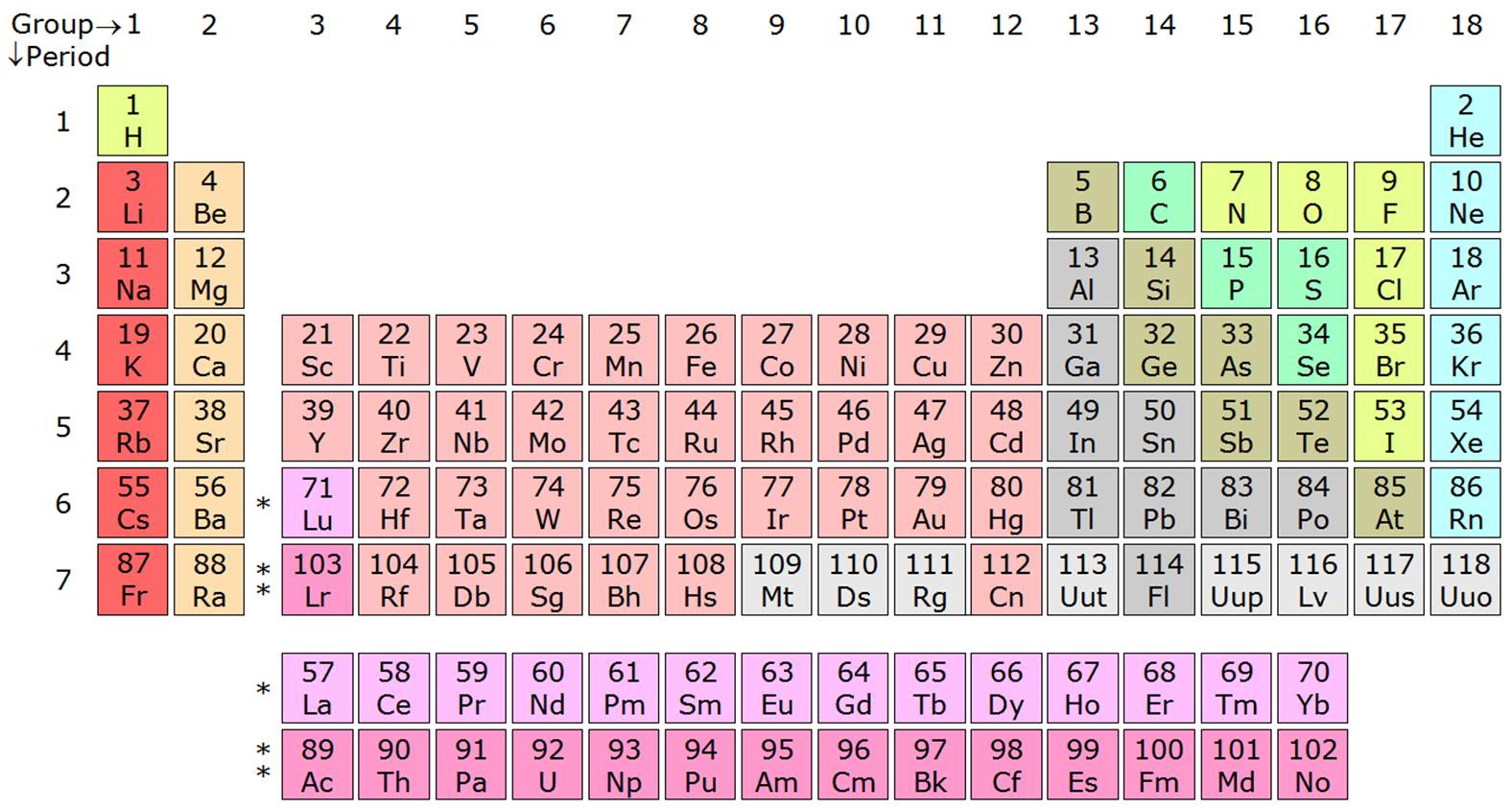
\): Summary of Periodic Trends in Atomic Properties. Thus, the periodicity of chemical properties is due to the periodicity of the valence electron configurations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
